
Antibody-Drug Conjugates Market to Witness Rapid Growth Across 7MM During the Study Period (2020–2034) as New Innovations Drive Therapy Advancements | DelveInsight
The antibody-drug conjugates market is fast evolving and is expected to grow significantly during the forecast period. The effectiveness of ADCs and checkpoint inhibitors in combating cancer is having a massive impact on the ADC R&D market; additionally, new and exciting uses of antibody-drug conjugates are also being actively researched.
/EIN News/ -- New York, USA, Feb. 20, 2025 (GLOBE NEWSWIRE) -- Antibody-Drug Conjugates Market to Witness Rapid Growth Across 7MM During the Study Period (2020–2034) as New Innovations Drive Therapy Advancements | DelveInsight
The antibody-drug conjugates market is fast evolving and is expected to grow significantly during the forecast period. The effectiveness of ADCs and checkpoint inhibitors in combating cancer is having a massive impact on the ADC R&D market; additionally, new and exciting uses of antibody-drug conjugates are also being actively researched.
DelveInsight’s Antibody-drug Conjugates Market Size, Target Population, Competitive Landscape & Market Forecast report includes a comprehensive understanding of current treatment practices, emerging ADCs, market share of individual therapies, and current and forecasted antibody-drug conjugates market size from 2020 to 2034, segmented into 7MM [the United States, the EU4 (Germany, France, Italy, and Spain), the United Kingdom, and Japan].
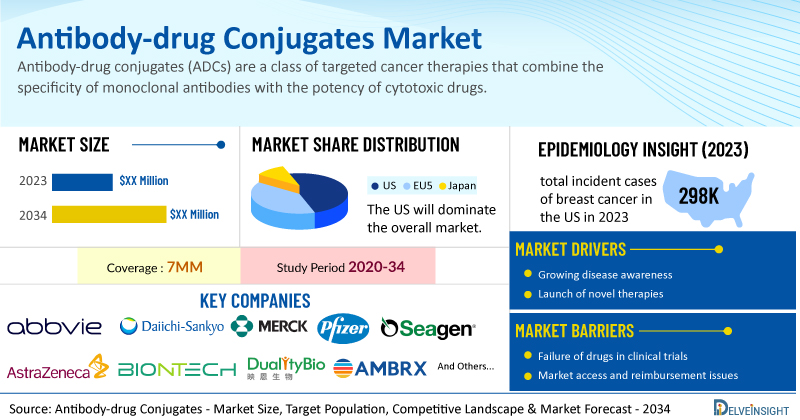
Key Takeaways from the Antibody-drug Conjugates Market Report
- As per DelveInsight’s analysis, the antibody-drug conjugates market is expected to grow significantly in the 7MM by 2034.
- The total sales of ENHERTU comprised nearly USD 1.4 billion in 2023 in the US.
- As per DelveInsight’s analysis, the total incident cases of breast cancer in the US comprised nearly 298K cases in 2023 and are projected to increase during the forecast period.
- Leading ADC companies such as AbbVie, Daiichi Sankyo, Merck, Pfizer, Seagen, AstraZeneca, BioNTech, Duality Biologics, Klus Pharma, Ambrx, Johnson & Johnson Innovative Medicine, GeneQuantum Healthcare (Suzhou), OnCusp Therapeutics, and others are developing novel ADCs that can be available in the antibody-drug conjugates market in the coming years.
- Some of the key antibody-drug conjugates in the pipeline include Telisotuzumab vedotin (ABBV-399), Patritumab deruxtecan (HER3-DXd), Ladiratuzumab vedotin, Dato-DXd, BNT323/DB-1303, A166, JNJ-0683 (ARX788), GQ1001, CUSP06, and others.
- In February 2025, the FDA awarded fast-track status to CUSP06, a CDH6-targeted ADC, for treating patients with platinum-resistant ovarian cancer.
- In January 2025, the FDA broadened the approval of ENHERTU to include individuals with hormone-positive, HER2-ultralow advanced breast cancer after undergoing endocrine therapy.
- In October 2024, Daiichi Sankyo announced that it has submitted a sNDA to Japan’s Ministry of Health, Labour and Welfare for ENHERTU for the treatment of adult patients with HER2 low or ultralow unresectable or recurrent breast cancer.
- In September 2024, AbbVie announced the submission of a Biologics License Application (BLA) to the US FDA for accelerated approval of telisotuzumab vedotin in adult patients with previously treated, locally advanced, or metastatic EGFR wild-type, non-squamous NSCLC with c-Met protein overexpression.
- In August 2024, ENHERTU was granted Breakthrough Therapy Designation in the US for the treatment of unresectable or metastatic hormone receptor-positive HER2 low or HER2 ultralow breast cancer patients.
Discover which therapies are expected to grab the ADCs market share @ Antibody-drug Conjugates Market and Competitive Landscape Report
Antibody-drug Conjugates Market Dynamics
The antibody-drug conjugate market is experiencing rapid growth, driven by advances in targeted therapies that offer highly specific treatments with reduced side effects compared to traditional chemotherapy. ADCs combine the targeting ability of monoclonal antibodies with the cytotoxicity of chemotherapy drugs, enabling them to precisely deliver drugs to cancer cells while minimizing damage to healthy tissues. This unique mechanism of action has resulted in a significant increase in ADC approvals for cancer treatments, especially for cancers that are difficult to treat with conventional therapies, such as breast cancer, lymphoma, and non-small cell lung cancer.
As the ADC market expands, pharmaceutical companies are heavily investing in research and development to enhance the effectiveness and safety profile of these therapies. Ongoing advancements in linkers, payloads, and antibody engineering are expected to further optimize ADCs for broader therapeutic indications. The market is also witnessing a surge in strategic collaborations and partnerships between biotech firms, major pharmaceutical companies, and contract research organizations (CROs), allowing for the acceleration of ADC product pipelines. These partnerships are critical in overcoming the high development costs and complex manufacturing processes associated with ADCs.
Despite the promising outlook, some challenges could hinder the growth of the ADC market. High production costs, limited scalability of manufacturing processes, and the risk of drug resistance in patients are some of the key obstacles. Furthermore, there is a need for better patient selection criteria, as not all patients will respond to ADC therapy. Regulatory hurdles also present a challenge, as the approval process for ADCs is complex due to their novel nature, requiring a thorough understanding of their pharmacokinetics and long-term safety profiles.
Antibody-drug Conjugates Treatment Market
ADC drugs combine the tumor-targeting ability of antibody therapies with the potent cell-killing effect of small molecule drugs, demonstrating remarkable clinical effectiveness in breast cancer, urothelial carcinoma, gastric cancer, and other areas. They offer additional treatment options for tumor patients who have not responded to multiple lines of therapy.
Some of the FDA-approved antibody-drug conjugates are KADCYLA, ADCETRIS, BESPONSA, MYLOTARG, LUMOXITI, POLIVY, PADCEV, TRODELVY, ENHERTU, BLENREP, ZYNLONTA, TIVDAK, and ELAHERE. In 2017, MYLOTARG was voluntarily removed from the market after follow-up trials did not confirm its clinical benefit and raised safety concerns, including a significant number of early deaths.
ENHERTU, developed by AstraZeneca and Daiichi Sankyo, is an antibody-drug conjugate targeting HER2 and combining trastuzumab with a topoisomerase inhibitor. It is approved for treating adult patients with unresectable or metastatic HER2-positive (IHC 3+ or ISH positive) breast cancer who have previously received an anti-HER2 treatment, as well as those with non-small cell lung cancer with a HER2 mutation.
ADCETRIS, developed by Seagen/Takeda, is a CD30-targeting monoclonal antibody linked to monomethyl auristatin E (MMAE), making it the second ADC to be introduced to the oncology market. It comprises the chimeric IgG1 antibody cAC10, which binds to human CD30, MMAE as the microtubule-disrupting agent, and a protease-sensitive linker that attaches MMAE to cAC10. ADCETRIS is approved in the US, Europe, and Japan for treating Hodgkin lymphoma and anaplastic large-cell lymphoma.
TRODELVY, developed by Gilead Sciences, is a conjugate consisting of a Trop-2-targeted antibody and a topoisomerase inhibitor, approved for the treatment of adult patients with unresectable locally advanced or metastatic triple-negative breast cancer who have undergone two or more previous systemic treatments, including at least one for metastatic disease. In 2023, TRODELVY generated sales of USD 1.06 billion.
Learn more about the FDA-approved ADCs @ Antibody-drug Conjugate Drugs
Key Emerging Antibody-drug Conjugates and Companies
Some of the emerging drugs are telisotuzumab vedotin, patritumab deruxtecan, ladiratuzumab vedotin, and camidanlumab tesirine, among others. Key players, such as ImmunoGen, Daiichi Sankyo, Seagen, Merck, ADC Therapeutics, CytomX, AbbVie, and others are involved in developing ADCs for indications such as non-small lung cancer, breast cancer, gynecological cancer, hematologic malignancies, and solid tumors.
Telisotuzumab vedotin is an ADC targeting cMet, currently under investigation for the treatment of NSCLC. The drug is being tested in the Phase III clinical trial TeliMET NSCLC-01 (M18-868) by AbbVie. In January 2022, AbbVie announced that the US FDA had granted Breakthrough Therapy Designation (BTD) for telisotuzumab vedotin to treat patients with advanced or metastatic EGFR wild-type, non-squamous NSCLC who have high levels of c-Met overexpression and whose disease has progressed after platinum-based therapy.
In September 2024, the company announced the submission of a Biologics License Application (BLA) to the US FDA for accelerated approval of telisotuzumab vedotin in adult patients with previously treated, locally advanced or metastatic EGFR wild-type, non-squamous NSCLC with c-Met protein overexpression.
Patritumab deruxtecan is an investigational ADC targeting HER3. Developed using Daiichi Sankyo's proprietary DXd ADC technology, it combines a fully human anti-HER3 IgG1 monoclonal antibody with topoisomerase I inhibitor payloads (an exatecan derivative, DXd) connected by cleavable tetrapeptide-based linkers. The drug received Breakthrough Therapy Designation (BTD) from the US FDA in December 2021 for treating patients with EGFR-mutated locally advanced or mNSCLC.
It is currently being tested in multiple Phase I, II, and III clinical trials. In October 2023, Daiichi Sankyo and Merck established a global partnership to develop and commercialize three DXd ADC candidates, including patritumab deruxtecan, ifinatamab deruxtecan (I-DXd), and raludotatug deruxtecan (R-DXd). In June 2024, the FDA issued a complete response letter regarding the approval application for third-line treatment of EGFR-mutated NSCLC based on Phase II trial results. In September 2024, the Phase III trial for second-line treatment of EGFR-mutated NSCLC (HERTHENA-Lung02) met its primary endpoint.
Seagen is a frontrunner in ADC research and its application in cancer treatment. Their antibody-drug conjugate product candidates include Ladiratuzumab vedotin, an ADC targeting LIV-1, which is currently undergoing Phase I/II clinical trials in combination with pembrolizumab for the first-line treatment of patients with unresectable locally advanced or metastatic triple-negative breast cancer.
The other ADCs in the pipeline include
- Dato-DXd: AstraZeneca/Daiichi Sankyo
- BNT323/DB-1303: BioNTech/Duality Biologics
- A166: Klus Pharma
- JNJ-0683 (ARX788): Ambrx/Johnson & Johnson Innovative Medicine
- GQ1001: GeneQuantum Healthcare (Suzhou)
- CUSP06: OnCusp Therapeutics, and others
The anticipated launch of these emerging ADCs are poised to transform the ADC market landscape in the coming years. As these cutting-edge therapies continue to mature and gain regulatory approval, they are expected to reshape the ADC market landscape, offering new standards of care and unlocking opportunities for medical innovation and economic growth.
To know more about ADCs clinical trials, visit @ Antibody-drug Conjugates in Development
Antibody-drug Conjugates Overview
Antibody-drug conjugates (ADCs) are a class of targeted cancer therapies that combine the specificity of monoclonal antibodies with the potency of cytotoxic drugs. The monoclonal antibody component of the ADC is engineered to recognize and bind to specific antigens found on the surface of cancer cells. Once bound, the ADC is internalized into the cancer cell, where the cytotoxic drug is released, delivering a highly targeted dose of chemotherapy directly to the tumor. This approach helps to minimize the systemic toxicity commonly associated with traditional chemotherapy, as the drug is delivered specifically to cancer cells rather than affecting healthy tissues.
The development of ADCs has led to significant advancements in cancer treatment, particularly for tumors that are resistant to conventional therapies. ADCs are designed to overcome some of the challenges in cancer therapy, such as off-target toxicity and drug resistance, by ensuring that the cytotoxic payload is only active inside cancer cells. However, the design and optimization of ADCs remain complex, involving careful selection of the monoclonal antibody, the cytotoxic drug, and the linker that connects them. Ongoing research is focused on improving the stability, potency, and selectivity of ADCs to enhance their therapeutic efficacy while minimizing side effects.
Antibody-drug Conjugates Epidemiology Segmentation
As per DelveInsight analysis, the total incident cases of TNBC in the United States comprised nearly 45K cases in 2023. The total number of incident cases of NSCLC in the 7MM were ~531K cases in 2023 which is expected to grow during the forecast period 2024–2034. Estimates show that the highest incident population of NSCLC is in the United States followed by Japan. The antibody-drug conjugates market report proffers epidemiological analysis for the study period 2020–2034 in the 7MM segmented into:
- Total Cases of Selected Indications for ADC
- Total Eligible Patient Pool for ADCs in Selected Indications
- Total Treated Cases in Selected Indications
Download the report to understand what epidemiologists are saying about ADCs patient trends in 7MM @ Antibody-drug Conjugates Patient Pool
| Antibody-drug Conjugates Report Metrics | Details |
| Study Period | 2020–2034 |
| Antibody-drug Conjugates Report Coverage | 7MM [the United States, the EU4 (Germany, France, Italy, and Spain), the United Kingdom, and Japan] |
| Key ADCs Companies | AbbVie, Daiichi Sankyo, Merck, Pfizer, Seagen, AstraZeneca, BioNTech, Duality Biologics, Klus Pharma, Ambrx, Johnson & Johnson Innovative Medicine, GeneQuantum Healthcare (Suzhou), OnCusp Therapeutics, and others |
| Key ADC in the Pipeline | Telisotuzumab vedotin (ABBV-399), Patritumab deruxtecan (HER3-DXd), Ladiratuzumab vedotin, Dato-DXd, BNT323/DB-1303, A166, JNJ-0683 (ARX788), GQ1001, CUSP06, and others |
Scope of the Antibody-drug Conjugates Market Report
- Antibody-drug Conjugates Therapeutic Assessment: Antibody-drug Conjugates current marketed and emerging therapies
- Antibody-drug Conjugates Market Dynamics: Conjoint Analysis of Emerging Antibody-drug Conjugates Drugs
- Competitive Intelligence Analysis: SWOT analysis and Market entry strategies
- Unmet Needs, KOL’s views, Analyst’s views, Antibody-drug Conjugates Market Access and Reimbursement
Discover more about ADC drugs in development @ Antibody-drug Conjugates Clinical Trials
Table of Contents
| 1. | Antibody-drug Conjugates Market Key Insights |
| 2. | Antibody-drug Conjugates Market Report Introduction |
| 3. | Executive Summary of Antibody-drug Conjugates |
| 4. | Key Events |
| 5. | Antibody-drug Conjugates Market Forecast Methodology |
| 6. | Antibody-drug Conjugates Market Overview at a Glance in the 7MM |
| 7. | Antibody-drug Conjugates: Background and Overview |
| 8. | Antibody-drug Conjugates Target Patient Pool |
| 9. | Antibody-drug Conjugates Marketed Drugs |
| 10. | Antibody-drug Conjugates Emerging Drugs |
| 11. | Seven Major Antibody-drug Conjugates Market Analysis |
| 12. | Antibody-drug Conjugates Market Access and Reimbursement |
| 13. | SWOT Analysis |
| 14. | KOL Views |
| 15. | Unmet Needs |
| 16. | Appendix |
| 17. | DelveInsight Capabilities |
| 18. | Disclaimer |
| 19. | About DelveInsight |
Related Reports
Antibody-drug Conjugates Competitive Landscape
Antibody-drug Conjugates Competitive Landscape – 2025 report provides comprehensive insights about the pipeline landscape, pipeline drug profiles, including clinical and non-clinical stage products, and the key ADC companies, including Sorrento Therapeutics, Ambrx, MacroGenics, SOTIO Biotech, Klus Pharma, BioAtla, BiOneCure Therapeutics, Shanghai Miracogen, ProfoundBio, ImmunoGen, Arcus Biosciences, CytomX Therapeutics, RemeGen, Bliss Biopharmaceutical, Aivita Biomedical, CoImmune, Zenith Epigenetics, Sutro Biopharma, Ambrx, Mythic Therapeutics, Shanghai Fudan-Zhangjiang Bio-Pharmaceutical, Byondis, MediLink Therapeutics (Suzhou), Oncomatryx Biopharma, MacroGenics, Shanghai Miracogen, Mirati Therapeutics, Ambrx, Orum Therapeutics, Silverback Therapeutics, Shanghai Fudan-Zhangjiang Bio-Pharmaceutical, Aivita Biomedical, Northwest Biotherapeutics, Daiichi Sankyo, Merck, Pfizer, GSK, AstraZeneca, Eisai, FOSUN PHARMA, LaNova, Mabwell Therapeutics, MBRACE THERAPEUTICS, MINGHUSI PHARMACEUTICALS, BioNTech , Bio-Thera, Corbus Pharmaceuticals, AbbVie, Multitude Therapeutics, Innovent, OnCusp Therapeutics, Simcere, Ymmunobio, Jiangsu Hengrui Medicine, among others.
Antibody-drug Conjugates in Oncology Competitive Landscape
Antibody-drug Conjugates in Oncology Competitive Landscape – 2025 report provides comprehensive insights about the pipeline landscape, pipeline drug profiles, including clinical and non-clinical stage products, and the key antibody-drug conjugates in oncology companies, including Seagen, Daiichi Sankyo, AstraZeneca, ADC Therapeutics, Abbvie, Gilead Sciences, Byondis, ImmunoGen, Merck, CytomX, Genmab, MacroGenics, among others.
Antibody Drug Conjugates Market
Antibody Drug Conjugates Market Insights, Competitive Landscape, and Market Forecast – 2032 report deliver an in-depth understanding of the market drivers, market barriers, and key ADC companies, including F. Hoffmann-La Roche Ltd, DAIICHI SANKYO COMPANY, LIMITED, Pfizer Inc., Gilead Sciences, Inc., ADC Therapeutics SA., GSK plc, Mersana Therapeutics., Astellas Pharma Inc., AstraZeneca, ImmunoGen Inc., among others.
Bispecific Antibody Competitive Landscape
Bispecific Antibody Competitive Landscape – 2025 report provides comprehensive insights about the pipeline landscape, pipeline drug profiles, including clinical and non-clinical stage products, and the key bispecific antibodies companies, including Janssen, Amgen, Akeso, Zymeworks, Roche, IGM Biosciences, MacroGenics, Provention Bio, Jiangsu Alphamab Biopharmaceuticals, Sichuan Baili Pharmaceutical, Regeneron Pharmaceuticals, Boehringer Ingelheim, among others.
Bispecifics/Trispecifics Market
Bispecifics/Trispecifics Market Forecast and Competitive Landscape – 2035 report deliver an in-depth understanding of the disease, historical and forecasted epidemiology, as well as the market trends, market drivers, market barriers, and key ADC companies, including Akeso, Summit Therapeutics, Zymeworks, BeiGene, Jazz Pharmaceuticals, Merus, Roche, GT Biopharma, Genor Biopharma, Numab Therapeutics, Sanofi, among others.
Oncology Conference Coverage Services
DelveInsight’s Oncology Conference Coverage Services offer a thorough analysis of outcomes from major events like ASCO, ESMO, ASH, AACR, ASTRO, SOHO, SITC, the European CAR T-cell Meeting, and IASLC. This detailed examination provides businesses with essential insights for competitive intelligence and market trend forecasting, supporting the formulation of future strategies.
Get in touch with us today to learn how we can provide AACR coverage exclusively for you at the AACR Meeting 2025
Other Business Consulting Services
Healthcare Competitive Intelligence
Healthcare Portfolio Management
About DelveInsight
DelveInsight is a leading Business Consultant and Market Research firm focused exclusively on life sciences. It supports pharma companies by providing comprehensive end-to-end solutions to improve their performance. Get hassle-free access to all the healthcare and pharma market research reports through our subscription-based platform PharmDelve.
Connect with us on LinkedIn|Facebook|Twitter

Contact Us
Shruti Thakur
info@delveinsight.com
+14699457679
www.delveinsight.com
Distribution channels: Healthcare & Pharmaceuticals Industry, Media, Advertising & PR, Science ...
Legal Disclaimer:
EIN Presswire provides this news content "as is" without warranty of any kind. We do not accept any responsibility or liability for the accuracy, content, images, videos, licenses, completeness, legality, or reliability of the information contained in this article. If you have any complaints or copyright issues related to this article, kindly contact the author above.
Submit your press release

